Intra-Trac®3 ADT
NOBIVAC® Intra-Trac®3 ADT is an intranasal vaccine that provides triple protection with Advanced Delivery Technology (ADT).
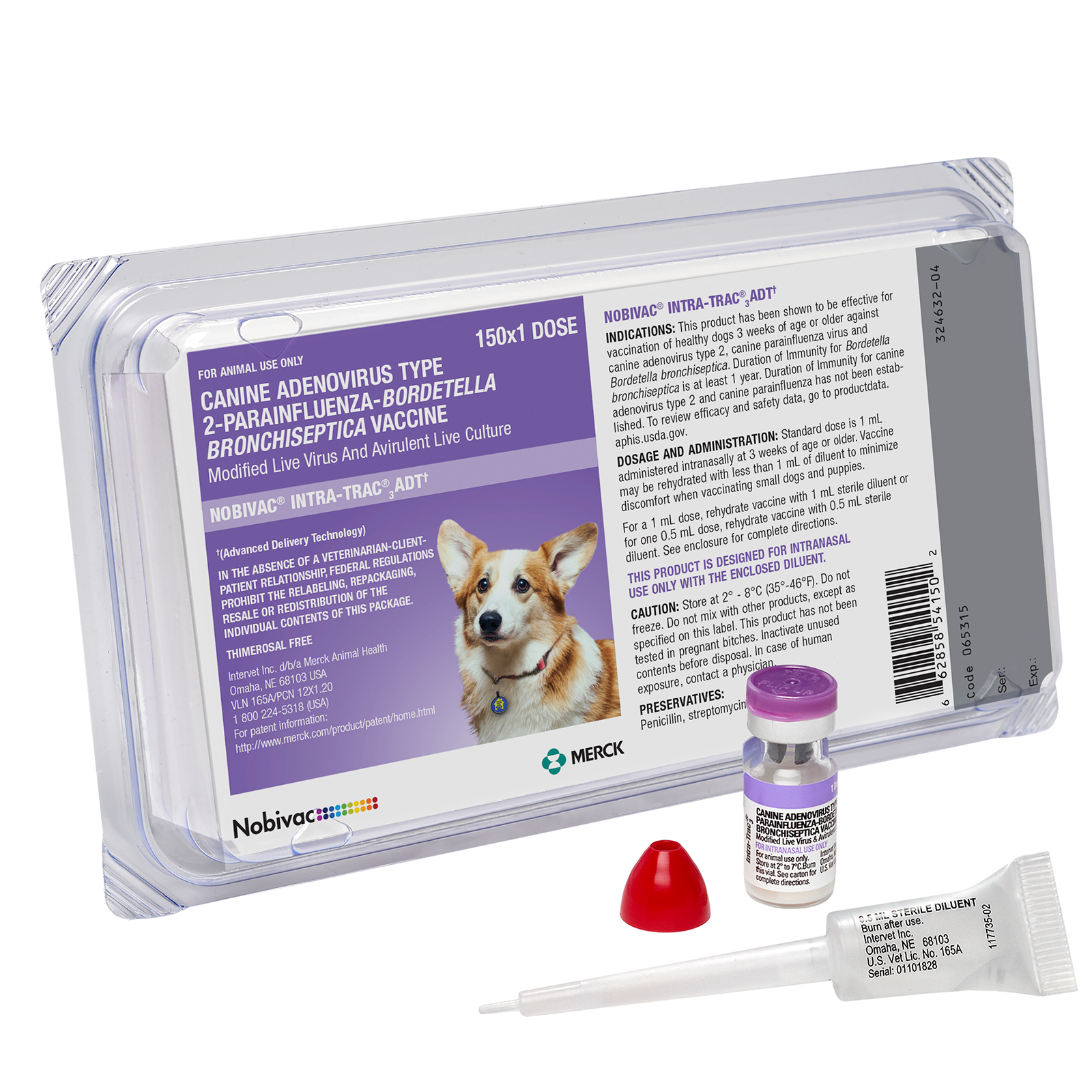
NOBIVAC® INTRA-TRAC®3 ADT PROVIDES PROTECTION AGAINST TRACHEOBRONCHITIS (“CANINE COUGH”)
Canine Adenovirus 2, Canine Parainfluenza Virus (modified live viruses), Bordetella Bronchiseptica (avirulent live culture).
- Protects against Bordetella, canine parainfluenza virus, and canine adenovirus type 2
- ADT eliminates the possibility of accidental injection
- Offers the superior benefits of intranasal administration
- Can be used in puppies as young as 3 weeks of age because maternal antibodies do not interfere with localized antibody response
- Requires only single-nostril administration—low dose of 0.5 mL
- Provides rapid protection beginning as early as 48 hours post-vaccination against
B. bronchiseptica14 - Proven 1-year duration of immunity for
B. bronchiseptica15
Indications:
This product has been shown to be effective for vaccination of healthy dogs 3 weeks of age or older against canine adenovirus type 2, canine parainfluenza virus and Bordetella bronchiseptica.
ADMINISTRATION AND DOSAGE
- Designed for intranasal use only with the enclosed applicators
- Using supplied nasal applicator, instill 1 dose (0.5 mL) into one or both nostrils
- Historically, annual revaccination has been recommended for this product. The need for this booster has not been established. Contact your veterinarian or manufacturer for more information on revaccination frequency.
- Available in two presentations: 25 x 1, 150 x 1 dose
ALSO AVAILABLE IN OTHER FORMULATIONS
DISEASE INFORMATION
Professional Resources and Educational Materials
Keep your clinic and staff informed and aware of diseases and outbreaks.

Pair Up for Protection
Infographic
When it comes to dogs in social environments, respiratory protection is essential. Use this conversation starter to discuss full respiratory protection with your clients.

Pair Up for Protection Social Media Kit
Digital Assets
This vaccination season, use these ready-to-share posts to educate pet parents about the importance of full canine respiratory protection.

Addressing CIV in Your Clinic
eBook
Tips and guidelines for your clinic to prevent and manage canine influenza.
No items to show.
References: