Nobivac® Lyme protects your dog against a growing threat.

Lyme disease is a nationwide threat1 that continues to spread, even in low-risk areas.
Lyme disease is a bacterial infection transmitted by ticks to dogs, other animals, and people. The disease can damage the heart, central nervous system, and kidneys, which can be fatal.2
Vaccination offers the best protection for your dog.
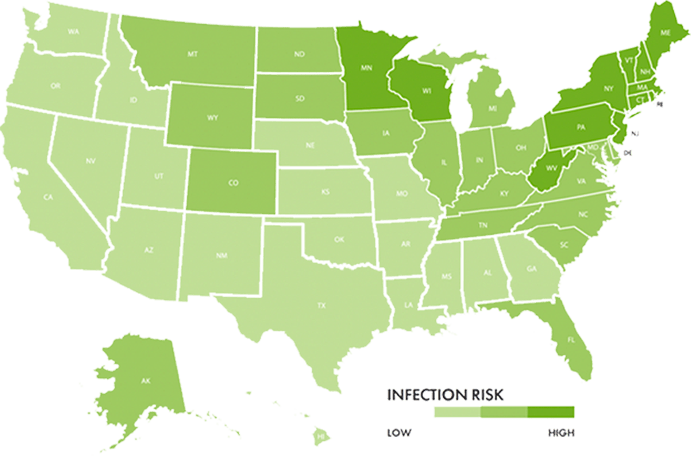
More than 30 states have a moderate-to-high prevalence of Lyme disease1

Why is Lyme disease spreading?
Once concentrated in the Northeastern US, tick populations are now spreading to the south and west into “lower-risk” areas like Ohio and North Carolina.
Factors causing tick populations to spread

Warmer temperatures

The migration of the
white-tail deer
Today, positive cases of Lyme disease have been reported in every state in the US.1
Lyme disease has increased by
~250%
NORTH CENTRAL STATES3
>320%
NORTHeastern STATES3
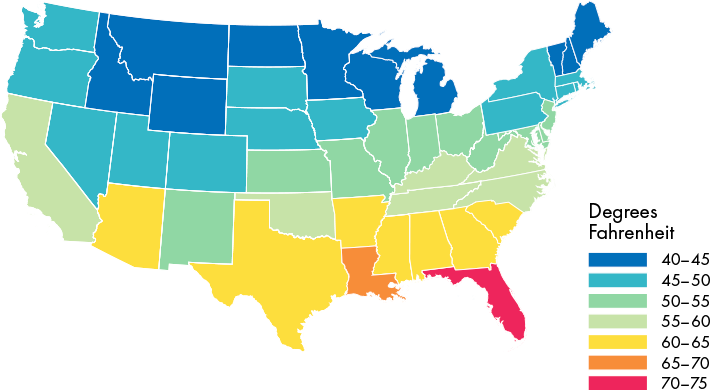

Average temperatures allow for year-round questing4
All of the Continental US report average annual temperatures above 40°F—warm enough to support tick activity all year long.5

Small size. Large threat.
A tick in the nymph stage is as small as a poppy seed. That’s why it is important to thoroughly check your dog for ticks.
Most Lyme disease in the US is spread by the deer tick, which is found primarily in the Northeast, mid-Atlantic region, and Upper Midwest.
The western black-legged tick is the primary source of Lyme disease transmission in the western states.6
Typically, ticks are most abundant in grassy, brushy, or wooded areas. However, their ability to attach themselves to humans and other animals means they can easily find their way inside, even latching onto dogs that spend all their time indoors.

A tick in the nymph stage is as small as a poppy seed.
The signs of Lyme disease
Only 5-10% of dogs show signs of Lyme disease.7 The most common signs include2:

Lethargy

Enlarged lymph
nodes

Shifting leg
lameness

Poor appetite

Joint swelling
Signs can occur as early as 2-5 months after infection, although it’s possible for them to develop much later.7
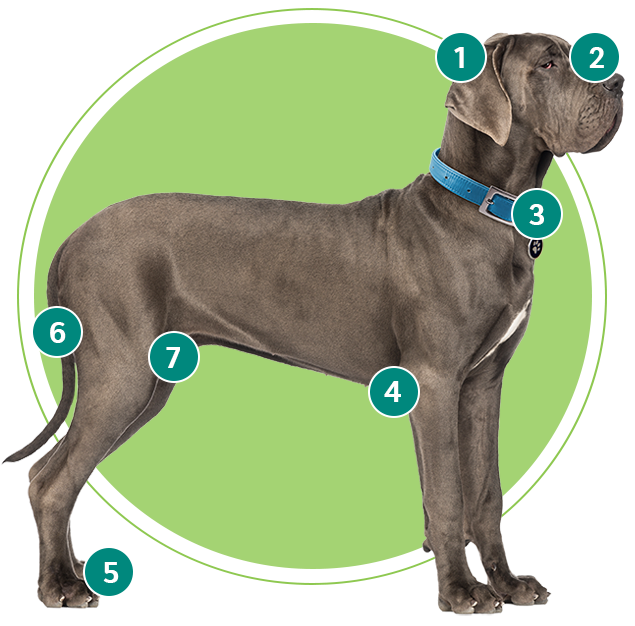
Where to check for ticks on your dog
- In and around the ears
- Around the eyelids
- Under the collar
- Under the front legs
- Between the toes
- Around the tail
- Between the back legs
The best protection is prevention
Dogs can be treated for Lyme disease with antibiotics, however, symptoms can return, requiring further treatment.2
To protect your dog from the bacteria that cause Lyme disease, your dog must be vaccinated against Lyme disease before it is bitten by an infected tick.2

of unvaccinated dogs in endemic areas tested positive for Lyme disease8

of adult ticks can be infected with the bacteria causing Lyme disease9
Vaccinating your dog may help keep them protected in case you don’t remove an infected tick in time.10

Protect with peace of mind
Lyme vaccines have been safely used to protect dogs since 1990.11
Side effects are rare, mild, and typcally resolve within a few days.12,13

References:
- Parasite prevalence map—ticks. Companion Animal Parasite Council website. Available at: https://capcvet.org/maps/#/2023/all-year/lyme-disease/dog/united-states. Accessed May 30, 2023.
- Staubinger, RK. Lyme disease (Lyme Borreliosis) in dogs. Available at: https://www.merckvetmanual.com/dog-owners/disorders-affecting-multiple-body-systems-of-dogs/lyme-disease-lyme-borreliosis-in-dogs. Accessed May 30, 2023.
- Kugeler KJ, Farley GM, Forrester JD, Mead PS. Geographic distribution and expansion of human Lyme disease, United States. Emerg Infect Dis. 2015;21:1455–1457.
- Average annual temperature for each US state. Current Results Weather and Science Facts website. Accessed May 30, 2023. https://www.currentresults.com/Weather/US/average- annual-state-temperatures.php
- Tick of the month: deer tick. YouTube page. Accessed April 19, 2021. https://www.youtube.com/watch?v=en9rk7wLfSk
- Lyme disease—transmission. Centers for Disease Control and Prevention website. Available at: https://www.cdc.gov/lyme/transmission/index.html. Accessed May 30, 2023.
- Lyme disease. Cornell University College of Veterinary Medicine website. Accessed May 30, 2023. https://www.vet.cornell.edu/animal-health-diagnostic-center/laboratories/serology-immunology/lyme-disease
- Levy SA. Use of a C6 ELISA test to evaluate the efficacy of a whole-cell bacterin for the prevention of naturally transmitted canine Bb infection. Vet Ther. 2002;3:420–424.
- Vogt NA. Lyme borreliosis in animals. Published March 2021. Updated October 2022. Accessed May 30, 2023. https://www.merckvetmanual.com/generalized-conditions/lyme-borreliosis/lyme-borreliosis-in-animals?redirectid=3170?ruleredirectid=30 1
- Lyme disease. Companion Animal Parasite Council website. Accessed May 30, 2023. https://capcvet.org/guidelines/lyme-disease/
- Canine Lyme disease vaccine introduced. United Press International website. Accessed May 30, 2023. https://www.upi.com/Archives/1994/10/05/Canine-Lyme-disease-vaccine-introduced/ 2060781329600
- Data on file. Merck Animal Health.
- Moore GE, Guptill LF, Ward MP, et al. Adverse events diagnosed within three days of vaccine administration in dogs. J Am Vet Med Assoc. 2005;227:1102–1108.