BANAMINE®
(flunixin meglumine injection)
Product Description
Banamine brand of flunixin meglumine is the pioneer injectable nonsteroidal anti-inflammatory drug approved for cattle and horses in the United States.
Each milliliter of BANAMINE contains flunixin meglumine equivalent to 50mg flunixin, 0.1mg edetate disodium, 2.5mg sodium formaldehyde sulfoxylate, 4.0mg diethanolamine, 207.2mg propylene glycol; 5.0mg phenol as preservative, hydrochloric acid, water for injection qs.
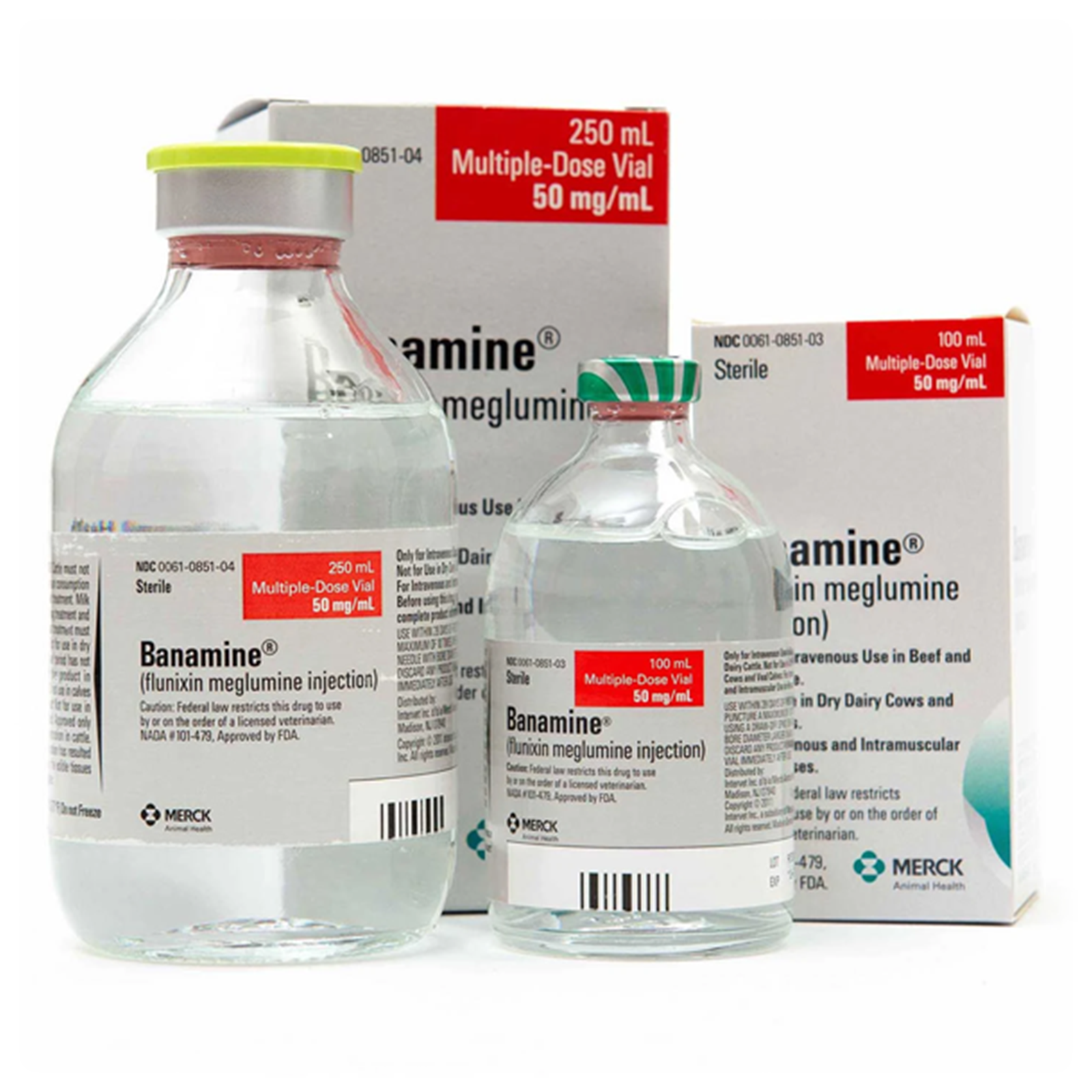


Indications
CATTLE: BANAMINE (flunixin meglumine injection) is indicated for the control of pyrexia associated with bovine respiratory disease, endotoxemia and acute bovine mastitis. BANAMINE is also indicated for the control of inflammation in endotoxemia.
HORSE: BANAMINE (flunixin meglumine injection) is recommended for the alleviation of inflammation and pain associated with musculoskeletal disorders in the horse. It is also recommended for the alleviation of visceral pain associated with colic in the horse.
Dosage
USE WITHIN 28 DAYS OF FIRST PUNCTURE AND PUNCTURE A MAXIMUM OF 10 TIMES. WHEN USING A DRAW-OFF SPIKE OR NEEDLE WITH BORE DIAMETER LARGER THAN 18 GAUGE, DISCARD ANY PRODUCT REMAINING IN THE VIAL IMMEDIATELY AFTER USE.
CATTLE: The recommended dose for control of pyrexia associated with bovine respiratory disease and endotoxemia and control of inflammation in endotoxemia, is 1.1 to 2.2 mg/kg (0.5 to 1 mg/lb; 1 to 2 mL per 100 lbs) of body weight given by slow intravenous administration either once a day as a single dose or divided into two doses administered at 12-hour intervals for up to 3 days. The total daily dose should not exceed 2.2 mg/kg (1.0 mg/lb) of body weight. Avoid rapid intravenous administration of the drug.
The recommended dose for acute bovine mastitis is 2.2 mg/kg (1 mg/lb; 2 mL per 100 lbs) of body weight given once by intravenous administration.
HORSE: The recommended dose for musculoskeletal disorders is 0.5 mg per pound (1 mL/100 lbs) of body weight once daily. Treatment may be given by intravenous or intramuscular injection and repeated for up to 5 days. Studies show onset of activity is within 2 hours. Peak response occurs between 12 and 16 hours and duration of activity is 24-36 hours.
The recommended dose for the alleviation of pain associated with equine colic is 0.5 mg per pound of body weight. Intravenous administration is recommended for prompt relief. Clinical studies show pain is alleviated in less than 15 minutes in many cases. Treatment may be repeated when signs of colic recur. During clinical studies approximately 10% of the horses required one or two additional treatments. The cause of colic should be determined and treated with concomitant therapy.
Contraindications
CATTLE: NSAIDs inhibit production of prostaglandins which are important in signaling the initiation of parturition. The use of flunixin can delay parturition and prolong labor which may increase the risk of stillbirth. Do not use BANAMINE (flunixin meglumine injection) within 48 hours of expected parturition. Do not use in animals showing hypersensitivity to flunixin meglumine. Use judiciously when renal impairment or gastric ulceration are suspected.
HORSE: There are no known contraindications to this drug when used as directed. Intra-arterial injection should be avoided. Horses inadvertently injected intra-arterially can show adverse reactions. Signs can be ataxia, incoordination, hyperventilation, hysteria, and muscle weakness. Signs are transient and disappear without antidotal medication within a few minutes. Do not use in horses showing hypersensitivity to flunixin meglumine.
Supplied
100-mL (50 mg/mL multi-dose vials)
250-mL (50 mg/mL multi-dose vials)
Storage
Store at or below 25°C (77°F) Do not Freeze.
Residue Warnings
Cattle must not be slaughtered for human consumption within 4 days of the last treatment. Milk that has been taken during treatment and for 36 hours after the last treatment must not be used for food. Not for use in dry dairy cows. A withdrawal period has not been established for this product in preruminating calves. Do not use in calves to be processed for veal. Not for use in horses intended for food. Approved only for intravenous administration in cattle. Intramuscular administration has resulted in violative residues in the edible tissues of cattle sent to slaughter.
Precautions
As a class, cyclo-oxygenase inhibitory NSAIDs may be associated with gastrointestinal and renal toxicity. Sensitivity to drug-associated adverse effects varies with the individual patient. Patients at greatest risk for renal toxicity are those that are dehydrated, on concomitant diuretic therapy, or those with renal, cardiovascular, and/or hepatic dysfunction.
Since many NSAIDs possess the potential to induce gastrointestinal ulceration, concomitant use of BANAMINE (flunixin meglumine injection) with other anti-inflammatory drugs, such as other NSAIDs and corticosteroids, should be avoided or closely monitored.
CATTLE: Do not use in bulls intended for breeding, as reproductive effects of BANAMINE (flunixin meglumine injection) in these classes of cattle have not been investigated. NSAIDs are known to have potential effects on both parturition (See Contraindications) and the estrous cycle. There may be a delay in the onset of estrus if flunixin is administered during the prostaglandin phase of the estrous cycle. NSAIDs are known to have the potential to delay parturition through a tocolytic effect. The use of NSAIDs in the immediate post-partum period may interfere with uterine involution and expulsion of fetal membranes. Cows should be monitored carefully for placental retention and metritis if BANAMINE is used within 24 hours after parturition.
HORSE: The effect of BANAMINE (flunixin meglumine injection) on pregnancy has not been determined. Studies to determine activity of BANAMINE when administered concomitantly with other drugs have not been conducted. Drug compatibility should be monitored closely in patients requiring adjunctive therapy.
Safety
CATTLE: No flunixin-related changes (adverse reactions) were noted in cattle administered a 1X (2.2 mg/kg; 1.0 mg/lb) dose for 9 days (three times the maximum clinical duration). Minimal toxicity manifested itself at moderately elevated doses (3X and 5X) when flunixin was administered daily for 9 days, with occasional findings of blood in the feces and/or urine. Discontinue use if hematuria or fecal blood are observed.
HORSE: A 3-fold intramuscular dose of 1.5 mg/lb of body weight daily for 10 consecutive days was safe.
No changes were observed in hematology, serum chemistry, or urinalysis values. Intravenous dosages of 0.5 mg/lb daily for 15 days; 1.5 mg/lb daily for 10 days; and 2.5 mg/lb daily for 5 days produced no changes in blood or urine parameters. No injection site irritation was observed following intramuscular injection of the 0.5 mg/lb recommended dose. Some irritation was observed following a 3-fold dose administered intramuscularly.
Adverse Reactions
In horses, isolated reports of local reactions following intramuscular injection, particularly in the neck, have been received. These include localized swelling, sweating, induration, and stiffness. In rare instances in horses, fatal or nonfatal clostridial infections or other infections have been reported in association with intramuscular use of BANAMINE (flunixin meglumine injection). In horses and cattle, rare instances of anaphylactic-like reactions, some of which have been fatal, have been reported, primarily following intravenous use.
Related Products
Contact
U.S. only: Merck Animal Health livestocktechsrvc@merck-animal-health.com or call 1-800-211-3573
For additional information, please see the product label.

For more cattle-friendly options, see the rest of the Merck Animal Health vaccine lineup.

Sign up to receive cattle health management insights,
industry news and more sent straight to your inbox.