D–CANINE DISTEMPER VIRUS (CDV)
- Onderstepoort-type strain in Nobivac® vaccines provides a high level of safety1
A–CANINE ADENOVIRUS TYPE 2 (CAV-2)
- Protection against CAV-1 and CAV-2 without the side effects associated with modified live CAV-1 vaccines1
P–CANINE PARAINFLUENZA VIRUS (CPIV)
- Systemic administration stimulates a strong IgG response1
PV-CANINE PARVOVIRUS (CPV)
- Protect against all known strains of Canine Parvovirus (CPV), including CPV-2C2,7,8
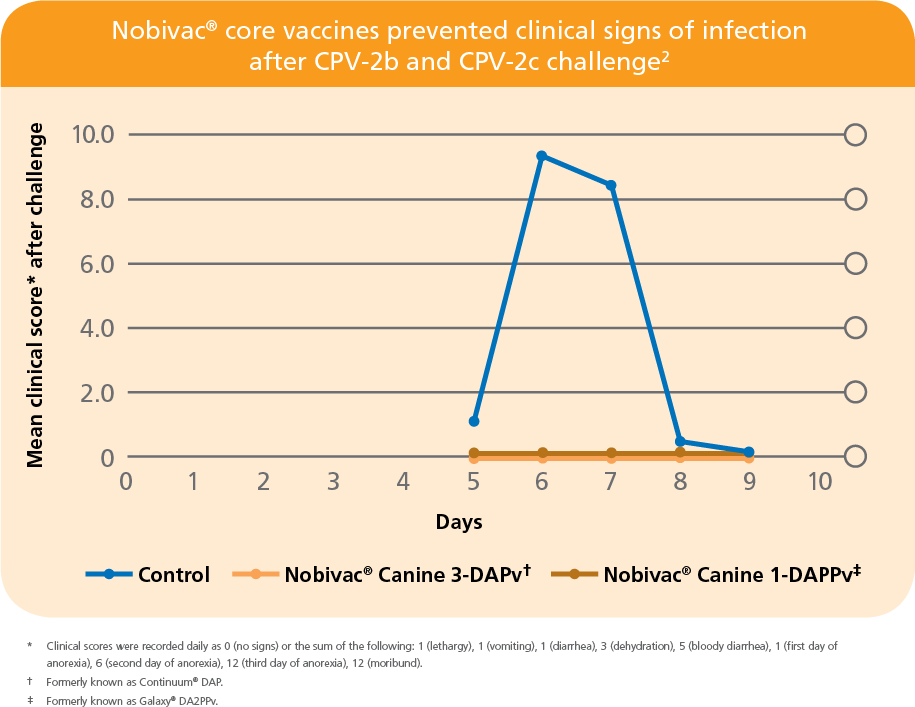
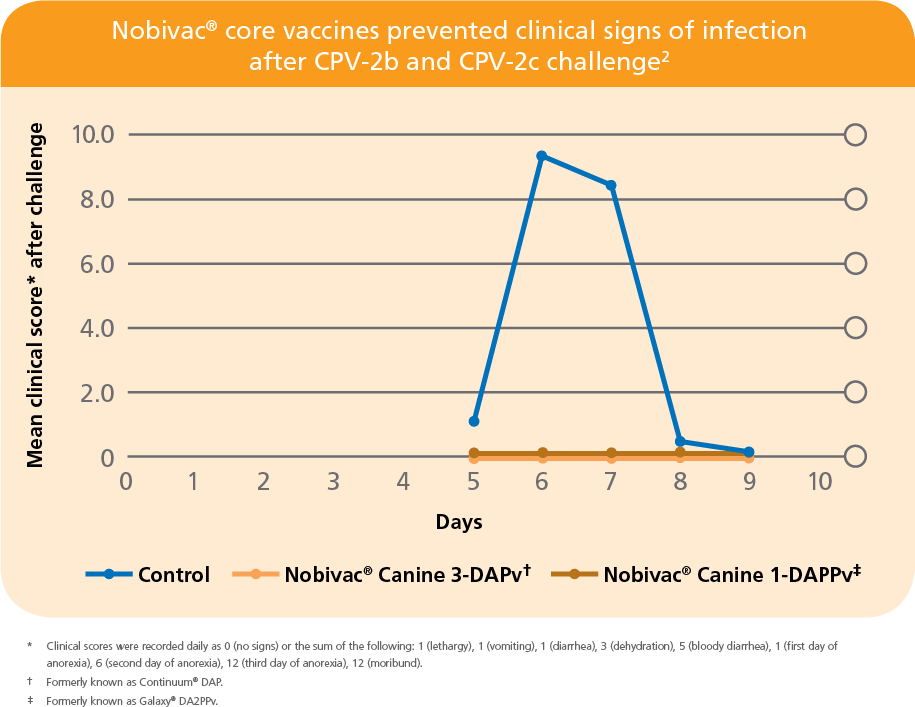
PROVEN IN AN INDEPENDENT STUDY BY LARSON AND SCHULTZ2
- No clinical signs of disease were seen in Nobivac-vaccinated puppies after challenge with mixed CPV-2b and CPV-2c strains2
- First published data showing protection against CPV-2c challenge2
- All vaccinated puppies were protected from disease
- All control puppies developed disease and 50% died or were euthanized