

Canine Influenza Virus
It only takes one dog infected by canine influenza virus (CIV) to spark an outbreak.
Overview | Transmission | Signs | Risk Factors | Outcomes | Spreading | Diagnosis | Vaccines | Resources
Disease Overview
Canine influenza is a relatively new disease caused by a “flu” virus. The first virus (H3N8) was thought to be of equine origin when found in Florida.
It is highly infectious and spreads rapidly. With initial signs being coughing, sneezing, nasal discharge and fever. Although it has low mortality rates, the morbidity rates are high.
CANINE INFECTIOUS FAST FACTS
- Canine Influenza spreads quickly and may cause serious illness to dogs, such as pneumonia.
- The H3N2 virus, identified in 2015, spread to half the continent in less than 6 months.
- CIV has now been identified in 46 states76.
- 20% of CIV-infected dogs do not exhibit clinical signs but still shed and spread the virus.36
TRANSMISSION
Canine influenza spreads the same as human flu:
- Direct contact (licking, nuzzling)
- Indirect (coughing or sneezing)
- Contaminated surfaces such as food and water bowls, cages, or human contact37,38
CLINICAL SIGNS38,39
- Sneezing
- Coughing (dry or moist)
- Low-grade fever
- Nasal discharge
- Lack of energy
- Loss of appetite
DOGS AT RISK
- Dogs that come from shelters, rescue centers, breeding kennels, or pet stores
- Boarding at a kennel or doggie daycare
- Visiting groomers, dog parks, or engaging with other dogs on a daily basis
- Dogs that participate in events/competitions
- Dogs that travel
- Dogs of veterinary staff that may be exposed through contaminated fomites
MORBIDITY THREATS
- The course of canine influenza is mild in most dogs, with signs developing less than 5 days after infection.
- Some dogs develop a more severe form with pneumonia and/or systemic illness.
- Approximately 80% of dogs, exposed to canine influenza virus H3N8, develop clinical disease.
- Up to 5-8% of dogs may progress to a more severe form of the condition – including fatalities.
- A subset of about 10 to 20% of dogs develop subclinical infection, but can still shed the virus and transmit disease to other dogs.
SPREADING DISEASE
- Shedding of CIV H3N8 begins about 24 hours after infection, tapers after a week, and typically disappears around day 10—just as the clinical signs of canine influenza are peaking.
- Shedding of CIV H3N2 has been shown to be longer – positives have been detected up to 24 days after the first positive on PCR, but may be intermittent.
DOGFLU.COM
Stop Canine Influenza in Your Community
DogFlu.com is a great resource to inform pet parents and professionals about the risks of canine influenza.
Heroes for Healthy Pets
Access free resources and protocols to keep veterinary clinics and pet businesses disease-free.
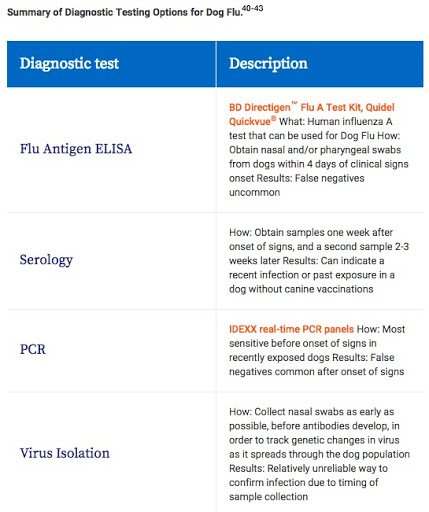
DIAGNOSIS
A definitive diagnosis of canine influenza can be made using laboratory testing, but timing of sample collection is critical and is related to the time of initial exposure and peak viral shedding.
Because of increasing prevalence, rapid spread, and the potential for up to 20% of dogs to have a severe disease course, veterinarians should examine further any dogs showing clinical signs.
A clinic can use multiple methods to test for both strains (CIV H3N8 and CIV H3N2).
Each of these tests can help with different readings, and several methods may be used depending on the stage of infection in the dog.40
In the video, Dr. Natalie Marks covers procedures and best practices for sampling and testing.
This can provide important information in making treatment decisions.
- A complete blood count (CBC) with differential, serum biochemistry profile, urinalysis, thoracic radiographs, and cultures can all provide helpful information. The CBC may be normal, or show mild leukopenia consistent with viral infection; however, leukocytosis (neutrophilia) with or without a left shift may indicate that the dog is developing pneumonia.
- Thoracic radiograph findings may range from mild bronchointerstitial infiltrates to consolidation of all lung lobes.
- Secondary bacterial infection may be identified by cultures performed on nasal swabs from dogs with purulent nasal discharge, or on transtracheal and endotracheal washes of dogs with pneumonia. Potential pathogens include a variety of gram-positive and gram-negative bacteria, including Staphylococcus spp., hemolytic and nonhemolytic Streptococcus spp., Pasteurella multocida, Klebsiella pneumoniae, Escherichia coli, and Mycoplasma spp.
- Antibiotic therapy can be targeted based on culture and sensitivity results.40
SEROLOGY
Serology to detect CIV-specific antibodies is the most reliable diagnostic test for confirmation of canine influenza infection.
- This testing should be performed in conjunction with other tests to confirm virus presence.
- CIV-specific antibodies can be detected in a hemagglutination inhibition assay as early as 7 days post infection; however, reliable detection occurs after 10 days of clinical signs.40,44
It is important to note that a negative antibody titer for serum samples collected before day 10 does not rule out CIV infection. In addition, because the presence of antibodies only indicates exposure but not necessarily active infection, it is necessary to compare an acute titer with a titer taken at least 2 weeks later (a convalescent titer) to confirm a four-fold or greater rise in antibody titer (seroconversion) in order to prove that there was a recent active infection.
Nasal and/or caudal pharyngeal swabs can also be used for detection of CIV nucleic acid by PCR tests.40 PCR testing is fast and inexpensive, and is now offered by several reference and university diagnostic laboratories.
In addition, PCR tests have high sensitivity and specificity, and are more likely to show positive results when there are low levels of virus present, such as in later stages of infection.
False-negative results can occur if the timing of sample collection is not appropriate.
Merck Animal Health Vaccines

NOBIVAC® CANINE FLU BIVALENT
The first licensed vaccine to protect your puppy or dog from both canine influenza strains H3N8 and H3N2. This product has been shown to be effective for vaccination of healthy dogs 7 weeks of age or older against canine influenza virus.
ALSO AVAILABLE
Professional Resources and Educational Materials
Keep your clinic and staff informed and aware of diseases and outbreaks.

The Infectious Disease Handbook
Stay on top of preventive care, and keep your business free from an outbreak, with this free handbook.

Nobivac® Social Media Kits
Digital Assets
Use these ready-to-share posts to educate pet parents and drive business to your clinic for preventative care.
No items to show.
Inform Pet Parents about Canine Influenza
Dog Flu Overview







